A suitable answer for every question.
General

To charge NiMH batteries safely and correctly, it is important to use a charger specifically designed for NiMH battery technology. This helps prevent overcharging, undercharging, and possible battery damage.
Slow charging is generally recommended, as it reduces heat generation and helps extend battery lifespan.
Well-known brands offering NiMH batteries include GP Batteries, Varta, and Duracell.
For the correct charging of NiMH batteries , it is essential to use a charger specifically designed for NiMH batteries to prevent overcharging or undercharging. Additionally, slow charging is recommended to avoid overheating and damage. Some brands that offer NiMH batteries include GP Batteries, Varta and Duracell.

A LiFePO4 battery looks the same as a conventional battery, but is lightweight, robust, and has high power. The LiFePO4 is a powerful battery that allows you to easily charge all your electronic devices. LiFePO4 batteries do not use toxic materials, thus reducing the risk of fire.
With a LiFePO4 battery, you can use more than 100% of its nominal capacity. In comparison, the usable capacity of a standard battery is between 30% and 50%.

Battery lifespan depends on several factors, including usage, charging behavior, depth of discharge, and ambient temperature.
To maximize battery life:
- Avoid frequent deep discharging
- Use the correct charger for the battery type
- Charge the battery according to the manufacturer’s recommendations
- Avoid excessive heat
- Store and use the battery at moderate temperatures
In most situations, an ambient temperature of around 20 °C is considered ideal. Higher temperatures can significantly reduce battery lifespan and performance over time. Under normal use and proper charging conditions, a battery will generally last considerably longer.

Proper battery storage helps extend battery lifespan. Storage recommendations differ per battery type:
- Li-ion and LiPo batteries: Store at approximately 50% charge in a cool, dark, and dry place. Never store fully charged or fully discharged batteries.
- Lead-acid batteries: Fully charged AGM and gel batteries can generally be stored unused for up to 6 months at around 20 °C ambient temperature.
- NiCd batteries: Best stored at approximately 40% charge to minimize capacity loss. Under proper conditions, NiCd batteries can often be stored for 3 to 5 years.

NiMH batteries should be charged using a charger specifically designed for NiMH battery technology. These chargers provide a controlled charging current and automatically stop charging once the battery is full, helping to prevent overcharging.
Fast chargers can recharge NiMH batteries within a few hours, but they generate more heat and place additional stress on the battery cells. Standard chargers charge more slowly but are generally better for battery lifespan and long-term performance.
To maximize battery life, it is recommended to fully charge the battery and avoid storing it empty for extended periods. Proper charging helps preserve battery capacity and ensures reliable operation over time.
Elfa supplies both NiMH batteries and reliable chargers for a wide range of applications.
Batteries

Alkaline batteries, lithium batteries, and specialist batteries such as air-alkaline and zinc-air alkaline. Alkaline batteries are often affordable and ideal for applications where power consumption is low. Lithium batteries can last up to seven times longer and offer higher peak power.
For more information about the different types of non-rechargeable batteries, please visit our page on non-rechargeable batteries. An example of a popular brand that produces high-quality non-rechargeable batteries is Procell.

These are the common abbreviations for battery systems. These terms are used universally and can also be found in this product catalog.
A round battery (see table) is indicated as round, and a flat battery as flat. The letter P is used for all batteries that are not round. Many chemical abbreviations can therefore include, in addition to an R, an F (flat) or a P.
B = Lithium carbon monofluoride (low drain)
BRC = Lithium manganese dioxide (high drain)
CR = Lithium manganese dioxide
ER = Lithium thionyl chloride
FR = Lithium iron disulfide
GR = Cadmium
KR = Alkaline
LR = Alkaline
MR = Mercury
PR = Zinc-air
SR = Silver oxide
An LR battery is therefore a round alkaline battery. A flat alkaline battery would therefore be designated as an LF battery, etc.

Alkaline batteries are by far the most commonly used type of primary battery. They are known for their high energy output, long shelf life, and reliable performance - even at relatively low temperatures.
Alkaline batteries are especially suitable for devices with low and continuous power consumption, such as remote controls, clocks, radios, and household electronics.
Want to learn more about alkaline batteries and other types of non-rechargeable batteries? Visit this page about primary batteries for additional information.

There are several types of button cell batteries, each designed for specific applications and performance requirements.
Silver oxide button cells
(commonly used in watches and cameras)
- Stable and constant voltage output
- Very low self-discharge: less than 5% per year
- Suitable for precision electronics
Alkaline button cells
(commonly used in electronic devices)
- Capable of delivering relatively high currents
- Voltage gradually decreases during use
- Self-discharge below 3% per year
Lithium button cells
(used in cameras and electronic devices)
- Extremely low self-discharge: less than 1% per year
- Long lifespan and high energy density
- Less suitable for very high continuous loads
Zinc-air button cells
(mainly used in hearing aids)
- Very high capacity
- Activated by exposure to air
- Self-discharge approximately 3% per year before activation
Mercury oxide button cells
(formerly used in electronic devices, hearing aids, and cameras)
- Delivered relatively high currents
- Self-discharge around 2% per year
- Contained approximately 15% mercury and were environmentally harmful
Mercury-containing batteries have no longer been produced since 1998 and have not been supplied by manufacturers since 1999.
For a limited number of applications, zinc-air batteries are available as replacement solutions.

The lifespan of a primary battery mainly depends on battery chemistry, ambient temperature, and the application’s energy consumption. Read more about primary battery lifespan.

To keep the battery status indication accurate, it is important to calibrate the battery periodically.
This is usually done by fully discharging the battery and then charging it completely back to 100%. During this process, the device recalibrates the battery status measurement, helping to improve the accuracy of the displayed charge level and overall battery performance.

Primary battery sizes are identified by both names and technical codes. The names are generally similar worldwide, although informal or local terms are also commonly used.
Battery codes can vary depending on the system used, such as:
- The international IEC code
- US battery codes
- Manufacturer-specific codes
Because of these different coding systems, battery markings on packaging can sometimes be confusing.
The IEC code is the most widely used international standard. In this system, the letters indicate the battery chemistry. For example:
- LR = Alkaline battery
- CR = Lithium battery
- SR = Silver oxide battery
The numbers in the code usually refer to the battery’s dimensions and size.


Because LiPo batteries contain lithium, they can pose a fire risk if stored incorrectly. It is therefore important to store them in a cool, dry, and dark environment away from direct sunlight and heat sources.
For additional safety, special LiPo Safe bags are available for storage and transport.
If you are not using a LiPo battery for an extended period, it is recommended to store it at approximately 60% charge (storage voltage). Many modern LiPo chargers include a dedicated storage mode for this purpose.

Yes. In addition to standard products, Elfa develops customized energy solutions tailored to specific customer applications and requirements. These include custom battery packs, lithium-ion systems, battery energy storage systems (BESS), and customer-specific electronics and lighting solutions.
Elfa combines more than 100 years of expertise with in-house production and assembly capabilities to develop solutions for sectors such as industry, OEM, defence, rail, telecom, emergency services, and renewable energy.
Customized solutions may include:
- Custom battery packs
- Lithium-ion battery systems
- Energy storage systems (BESS)
- Emergency power solutions
- Custom lighting solutions
- Customer-specific electronics integration
Elfa also advises on total cost of ownership, battery lifespan, and system optimization during the development process.

There are several types of rechargeable batteries, each with its own characteristics and applications:
- NiMH batteries (Nickel Metal Hydride)
Commonly used in consumer electronics such as remote controls, flashlights, and portable devices. They offer relatively high capacity and are widely used as rechargeable AA and AAA batteries. - NiCd batteries (Nickel-Cadmium)
Less common today because of their lower capacity and sensitivity to the so-called “memory effect”. Still used in certain industrial applications, cordless phones, and tools. - Lithium-ion batteries (Li-ion)
Widely used in smartphones, laptops, electric vehicles, and industrial applications due to their high energy density, long lifespan, and low self-discharge. - Lithium polymer batteries (LiPo)
A variation of Li-ion technology with a lightweight and compact design. Often used in drones, tablets, smartphones, and other portable electronics. - Lead-acid batteries
Commonly used in vehicles, backup systems, boats, and industrial equipment. Known for their ability to deliver high current and relatively low cost. - Lithium iron phosphate batteries (LiFePO4)
Known for their long cycle life, high safety level, and thermal stability. Frequently used in energy storage systems, electric vehicles, and industrial applications. - Zinc-carbon rechargeable batteries
Less commonly used today because of their lower capacity and shorter lifespan compared to modern battery technologies.
Choosing the right rechargeable battery depends on the application, required capacity, operating conditions, and expected lifespan. Proper charging and maintenance are essential for optimal performance and battery longevity.

Rechargeable batteries offer many advantages, but there are also some disadvantages to consider.
One of the main drawbacks is that rechargeable batteries can suddenly stop working when they are empty. In devices such as cameras, flashes, or power tools, this can happen unexpectedly. Alkaline batteries usually lose voltage more gradually, giving more warning before they stop functioning completely.
Rechargeable batteries are also generally more expensive to purchase than alkaline batteries, partly because a suitable charger is required as well.
Nickel-cadmium (NiCd) batteries contain cadmium, a heavy metal that is harmful to the environment. However, these batteries can be recycled effectively. NiCd batteries are also sensitive to the so-called “memory effect”, meaning they should ideally be fully discharged before recharging to maintain capacity.
Nickel-metal hydride (NiMH) batteries largely eliminate this problem and can be recharged at almost any time without significant memory effect.

Lithium batteries are a special type of battery with several important advantages compared to traditional battery technologies.
- Ideal for applications that require high peak power
- On average, they last up to 7 times longer than alkaline batteries
- Suitable for temperatures ranging from -20 °C to +40 °C
- Extremely low self-discharge: only around 1% capacity loss per year
- High operating voltage and high energy density
- Lightweight compared to many other battery chemistries
Lithium is a lightweight metal with an exceptionally high energy capacity of approximately 3.86 Ah per gram. When combined with materials such as manganese or sulfur, it creates batteries with high energy output, low weight, long shelf life, and reliable performance under extreme conditions.
Lithium batteries are used in a wide variety of applications, including:
- Mobile phones and portable electronics
- Microprocessors and IoT devices
- Medical equipment
- Industrial applications
- Oil and gas industry
Read more about lithium batteries and their applications here.

NiMH (Nickel Metal Hydride) batteries offer several advantages over older rechargeable battery technologies such as NiCd.
- Higher energy density than NiCd batteries
- Less sensitive to the memory effect
- More environmentally friendly because they contain fewer harmful metals
- Suitable for rechargeable AA and AAA applications
- Widely used in consumer electronics and portable devices
Well-known brands that produce NiMH batteries include Varta, Duracell, and GP Batteries.

Rechargeable batteries offer several important advantages over non-rechargeable alkaline batteries:
- Long lifespan - Many rechargeable batteries can be recharged hundreds or even thousands of times, making them highly durable.
- Cost-effective - Although the initial purchase price is higher, rechargeable batteries and chargers quickly pay for themselves when batteries are used regularly.
- More environmentally friendly - Because rechargeable batteries are reused many times, far fewer disposable batteries end up as waste. This helps reduce environmental impact and supports more sustainable energy use.
- Reliable brands - Well-known brands such as Saft, Tadiran, and Varta produce high-quality rechargeable batteries that are often available directly from Elfa stock.
Before switching to rechargeable batteries, it is important to verify whether the specific application is suitable for rechargeable battery technology.

Alkaline batteries are the most commonly used type of primary battery and offer several important advantages:
- Low purchase cost
- Reliable energy output, even at relatively low temperatures
- Low self-discharge for long shelf life
- Suitable for devices with low to moderate power consumption
Well-known brands offering alkaline batteries include Procell, Panasonic, Energizer, and GP Batteries.

Nickel Metal Hydride (NiMH) batteries are rechargeable batteries known for their reliability and environmentally friendly characteristics.
NiMH batteries are widely used as an alternative to NiCd batteries, which are restricted in many portable applications due to environmental regulations.
Key advantages of NiMH batteries include:
- High energy capacity
- No cadmium content
- Rechargeable hundreds of times
- Minimal memory effect
- Stable performance across many applications
NiMH batteries are commonly used in:
- Flashlights and headlamps
- Security and emergency equipment
- Consumer electronics
- Industrial applications
Thanks to their strong price-performance ratio, NiMH batteries are a practical and sustainable choice for professional use.
Elfa supplies a wide range of NiMH batteries as well as compatible standard and fast chargers.

Air-alkaline batteries are a special type of primary battery that uses oxygen from the air to generate energy. Because of this technology, they offer a high energy density with relatively low weight.
Advantages of air-alkaline batteries include:
- High energy density
- Low cost
- Environmentally friendly characteristics
- Long shelf life before activation
Once activated by exposure to air, the battery starts discharging immediately and the chemical process cannot be fully stopped.
Air-alkaline batteries are commonly used for:
- Railway signal lighting
- Road signal lighting
- Marine and buoy lighting
- Telecommunications
- Parking meters
- Electric fencing systems

Nickel cadmium batteries and niNickel Metal Hydride (NiMH) batteries are considered the successor to Nickel-Cadmium (NiCd) batteries and offer several advantages.
The main differences are:
- NiMH batteries can offer up to twice the capacity of NiCd batteries
- NiCd batteries are more sensitive to the “memory effect”
- NiMH batteries require less maintenance and care
- NiMH batteries are generally more environmentally friendly because they contain less harmful material
Because of these advantages, NiMH batteries are now more commonly used in many rechargeable battery applications.

There are several important differences between lithium and alkaline batteries:
- Lithium batteries generally last up to 7 times longer than alkaline batteries
- Alkaline batteries are usually more affordable
- Lithium batteries are often used in professional and industrial applications
- Alkaline batteries are mainly used in consumer devices such as remote controls and radios
- Lithium batteries perform better under extreme temperatures and high energy demand
Well-known lithium battery brands include Saft, Tadiran, and GP Batteries. Popular alkaline battery brands include Procell and Panasonic.

Alkaline button cell batteries provide relatively high currents thanks to their initial voltage of 1.5 V. However, the voltage drops relatively quickly during use.
Because of this, alkaline button cells are less suitable for precision devices such as cameras and measuring equipment, where stable voltage is important.

Nickel-cadmium (NiCd) batteries are rechargeable batteries originally invented in 1899 by Waldmar Jungner.
Key characteristics of NiCd batteries include:
- Very robust and durable construction
- Fast charging capability with relatively low stress on the battery
- Good performance at low temperatures
- Suitable for demanding industrial applications

A coin cell battery is a small, round battery with a diameter that is usually larger than its height. Because many of these batteries are very compact and resemble a button in shape, they are also commonly referred to as button cell batteries.

LiPo stands for Lithium Polymer or Lithium-Ion Polymer. A LiPo battery is a lightweight rechargeable battery with high capacity and a long lifespan.
LiPo batteries are widely used in:
- Smartphones
- Laptops
- Drones
- RC vehicles
- Portable electronics
A LiPo battery consists of one or more cells with a nominal voltage of 3.7 V per cell. When fully charged, each cell reaches approximately 4.2 V.
The number of cells is indicated with an “S” rating, such as:
- 2S LiPo
- 3S LiPo
- 4S LiPo
Thanks to their high power output and low weight, LiPo batteries are especially suitable for applications where performance and compact size are important.
To maximize lifespan and safety, LiPo batteries must be charged, discharged, and stored correctly.

NiMH batteries are known for their relatively high cycle life. On average, a NiMH battery can handle approximately 700 to 1,000 charge cycles.
If the battery is repeatedly deeply discharged, performance may start to decline after around 200 to 300 cycles.
In general, NiMH batteries have an expected lifespan of approximately 2 to 3 years, depending on usage and charging conditions.

The OPEN Foundation (Organization for Producer Responsibility for E-waste Netherlands) is responsible for the collection and recycling of discarded electrical equipment and used batteries in the Netherlands. The organization continues the activities previously carried out by Stibat.
As a non-profit organization, Stichting OPEN helps producers and importers comply with legal recycling obligations for batteries and electronic waste. Since 1995, regulations have required batteries to be collected and recycled responsibly. This system is financed through a recycling contribution paid per battery.
Used batteries should never be disposed of with regular household waste because they may contain environmentally harmful substances such as mercury, lead, or cadmium. By collecting batteries separately, valuable raw materials such as nickel, steel, and zinc can be safely recovered and reused in new products.
The collection system includes:
- Household batteries
- Button cells
- Rechargeable batteries
- Batteries from phones, cameras, and power tools
Through large-scale recycling efforts, millions of kilograms of reusable materials have already been recovered in the Netherlands, reducing environmental impact and conserving natural resources.
Read more about battery recycling and the OPEN Foundation here.

A zinc-air battery is activated only after the protective sticker is removed. Once opened, oxygen from the air enters the battery through small holes in the housing and activates the chemical process.
Because part of the active material comes from the surrounding air, more internal space is available for energy capacity. This results in:
- High energy density
- Long operating time
- Stable discharge performance
Air-alkaline batteries work according to a similar principle.

Lithium button cell batteries have a very long lifespan and can often last up to 10 years, even with little or no use.
They are characterized by extremely low self-discharge — typically only around 1% capacity loss per year — making them ideal for long-term applications.
Lithium button cells are commonly used in:
- Watches
- Medical devices
- Car keys
- Electronic devices
- Backup memory applications
Procell is one example of a brand that offers lithium button cell batteries.
Lithium batteries last a long time, sometimes up to ten years, even when they are not or barely used. Each year, self-discharge is only 1% of the capacity. Procell is an example of a brand that offers button batteries.

Both NiMH and NiCd batteries are suitable for applications with relatively high power consumption. However, there are several important differences between the two technologies.
NiCd batteries are known for their robustness and are often used in power tools such as drills. They perform well under demanding conditions and at low temperatures.
A major disadvantage of NiCd batteries is that they contain cadmium, a toxic and environmentally harmful heavy metal. Because of environmental regulations, the use of NiCd batteries in many portable applications has been restricted, and they are increasingly being replaced by NiMH batteries.
NiMH batteries offer a higher energy density and larger capacity, allowing devices to operate longer on a single charge. They are commonly used in portable electronics such as laptops, flashlights, and consumer devices.
Advantages of NiMH batteries include:
- Higher capacity
- Longer runtime
- No toxic cadmium
- Reduced memory effect compared to NiCd
One limitation of NiMH batteries is that they are generally less resistant to extreme heat and cold. For optimal lifespan and performance, NiMH batteries should preferably be stored at cool ambient temperatures between 1 °C and 25 °C.

No, standard alkaline batteries are not rechargeable. Although rechargeable alkaline variants do exist, they have several important disadvantages.
Disadvantages of rechargeable alkaline batteries
- They require a special charger
- Charging them in a NiMH or NiCd charger is unsafe and may cause leakage or explosion
- Their capacity decreases after every charge cycle
- They can only be recharged a limited number of times
- The efficiency is relatively low compared to real rechargeable batteries
- Once the voltage drops below a critical level, recharging is no longer possible
The only real advantage is their slightly higher initial voltage compared to NiMH batteries. However, during use this voltage drops quickly.
For most applications, true rechargeable batteries such as NiMH batteries are the better and safer choice.

Yes, NiMH batteries can be charged quickly. However, frequent fast charging may reduce battery lifespan and capacity over time.
For optimal performance and longevity, slower charging is generally recommended.

No, NiMH batteries should only be charged with a charger specifically designed for NiMH technology. Using the wrong charger may damage the battery and reduce its lifespan.

In many applications, a 1.2 V rechargeable battery can be used without problems.
Although alkaline batteries start at 1.5 V, their voltage quickly drops during use and can eventually fall below the voltage level of a rechargeable battery. Rechargeable batteries such as NiMH batteries provide a much more stable voltage during discharge.
Because of this stable voltage curve, rechargeable batteries perform well in many everyday devices.

A rechargeable battery stores electrical energy and can be recharged and reused after it has been discharged - similar to a car battery.
With proper use and charging, rechargeable batteries can often be charged hundreds or even thousands of times.
Rechargeable batteries such as NiMH batteries provide a relatively stable voltage of around 1.2 V during use. This means they deliver consistent performance almost until they are empty, after which the voltage drops quickly and the battery needs recharging.
Well-known rechargeable battery brands available at Elfa include Saft, Tadiran, and GP Batteries.

It is recommended to fully charge a lithium-ion battery to 100% only during the first charge cycle. For daily use, it is better to disconnect the battery from the charger before it reaches full charge.
Avoiding continuous full charging can help extend the lifespan of a lithium-ion battery.

LiPo batteries require a special charging method and should only be charged with a dedicated LiPo charger. During charging, the current and voltage must be carefully controlled to prevent overcharging and damage.
A suitable LiPo charger uses a balancing function that monitors and balances the individual cells during charging. Once the maximum cell voltage is reached, the charger automatically reduces and eventually stops the charging current.
In addition to the positive and negative terminals, LiPo chargers also use a balancing connector to safely charge each cell evenly. This ensures controlled, balanced, and safe charging of the LiPo battery pack.

A battery can be tested using a multimeter or a dedicated battery tester.
Simple battery testers are often used for AA and AAA batteries and indicate whether the battery still contains sufficient charge. Basic models typically use an LED indicator, while more advanced testers measure the actual battery voltage.
A multimeter can also be used to check battery voltage. For example, if a 1.5 V battery measures only 1.2 V, this may indicate that the battery capacity has significantly decreased.

NiMH batteries are best stored in a cool, dry environment and preferably in a partially charged condition. This helps minimize self-discharge and can extend battery lifespan.

Yes, there are various types of rechargeable button cell batteries. These batteries are often integrated into devices and are commonly used in applications such as backup systems, medical devices, and small electronics.
Rechargeable button cells are less commonly available in regular stores because demand is relatively limited compared to standard disposable button batteries.

Yes, NiMH batteries are generally considered more environmentally friendly than NiCd batteries because they contain fewer toxic and harmful metals.
To minimize environmental impact, it is important to recycle NiMH batteries properly at designated collection points.

Lithium Iron Phosphate (LiFePO4) batteries are known for their very low fire risk and high level of safety.
Compared to other lithium-ion technologies, LiFePO4 cells are thermally very stable and far less likely to overheat, ignite, or explode. They can withstand high temperatures and remain relatively safe even in cases of improper charging or mechanical damage.
Because of this stability, LiFePO4 batteries are widely used in applications where reliability and safety are critical.
In short: LiFePO4 batteries offer energy storage with a very low fire hazard and a high degree of operational safety.

Lithium-ion (Li-ion) batteries can store more energy than NiCd and NiMH batteries and offer a very high energy density — up to approximately 140 Wh/kg.
The actual lifespan of a Li-ion battery depends on factors such as:
- The application
- Number of charge and discharge cycles
- Charging behavior
- Operating temperature
To maximize battery lifespan:
- Avoid fully charging the battery continuously
- Preferably keep the charge level between 20% and 80%
- Prevent deep discharge
- Store the battery at around 40% charge when not used for long periods
- Store the battery in a cool environment below 25 °C
With proper use and storage, lithium-ion batteries can achieve a long operational lifespan and stable performance.

A battery recycling fee is a legally required environmental contribution for products that contain a battery or are supplied with one. This fee helps finance the collection, processing, and recycling of used batteries in accordance with environmental regulations.
The recycling fee applies not only to loose batteries, but also to products with integrated or included batteries, such as flashlights and portable electronics.
Where can batteries be recycled?
- Consumers can return used batteries at municipal chemical waste collection points or approved collection locations
- Companies can arrange collection through certified recycling and waste processing companies
Proper collection and recycling ensure that harmful substances are processed safely and valuable raw materials can be recovered and reused.
Read more about the current battery recycling fees here.

The capacity — and therefore the runtime — of a battery is expressed in milliampere-hours (mAh). This indicates how much current a battery can deliver over a certain period of time.
mAh stands for:
- mA = milliampere (one thousandth of an ampere)
- h = hours
The higher the mAh value, the longer the battery can generally power a device before recharging or replacement is required.

Internal resistance indicates how easily a battery can deliver electrical energy. In addition to voltage (V), internal resistance — expressed in ohms (Ω) — plays an important role in battery performance.
A battery with low internal resistance can deliver energy more efficiently and handle higher current demands better. Batteries with higher internal resistance experience more energy loss and are generally less suitable for high-power applications.
Battery types with relatively low internal resistance include:
- Nickel-Cadmium (NiCd)
- Nickel Metal Hydride (NiMH)
- Alkaline
- Lithium Photo
- Mercury Oxide
Battery types with relatively higher internal resistance include:
- Lithium primary batteries
- Zinc-air batteries
- Silver oxide batteries
- Zinc-carbon batteries

Button batteries, also known as button cells, are mainly used in small electronic devices that require compact and reliable power sources.
Common applications include:
- Watches
- Cameras
- Hearing aids
- Calculators
- Small electronic games
- Car alarms and remote keys
Because of their compact size, button cells are ideal for devices where space is limited.

Silver oxide batteries are commonly used in watches because they provide a high and very stable voltage over a long period of time. This stable voltage ensures reliable and accurate timekeeping, which is essential for watch performance.

NiMH batteries naturally have a relatively high self-discharge rate compared to some other battery technologies. This means they gradually lose charge even when not being used.
Self-discharge can be reduced by:
- Storing the batteries in a cool, dry environment
- Keeping them partially charged during storage
- Using and recharging them regularly
Proper storage and maintenance help preserve battery performance and lifespan.

On most coin cell batteries, the negative terminal is the flat top side, while the positive terminal is the slightly wider bottom side.
The top side usually displays the brand name and battery type number and is typically the side facing upward during installation.

Yes. ELP NiCd batteries are still available and have been specifically developed for use in emergency lighting systems. These batteries fully comply with the applicable regulations and are classified as industrial batteries in accordance with Regulation (EU) 2023/1542 of July 12, 2023.

Yes, button cell batteries can operate under a wide temperature range. Depending on the battery type, the allowable temperatures can range from approximately -55 °C to +125 °C.
For optimal performance and lifespan, most button cell batteries function best at temperatures between +10 °C and +35 °C.
Procell is one example of a brand offering button cell batteries that are available from Elfa.

Yes. In general, the higher the battery capacity, the longer the battery can power a device. Battery capacity is expressed in ampere-hours (Ah) or milliampere-hours (mAh).
The capacity is usually indicated on both the battery and its packaging.
Using a battery with a higher capacity is generally not harmful and simply provides longer runtime. However, it is important to always use the correct voltage specified for the device. Using the wrong voltage may damage the equipment.

A salt water battery works by generating energy through the interaction between salt water and fresh water in separate compartments inside the battery system.
During discharge, the contact between these liquids creates an electrical current. During charging, the salt water and fresh water are separated again.
Salt water batteries often use materials such as manganese oxide and carbon-titanium phosphate as part of the electrochemical system.
Advantages of salt water batteries include:
- Environmentally friendly technology
- Easy maintenance
- High safety level
- Suitable for household energy storage
- Long lifespan of up to approximately 20 years
Because they do not use highly flammable materials, salt water batteries are considered a safe and sustainable energy storage solution.

A battery generates electrical energy through internal chemical reactions. It consists of a positive terminal, a negative terminal, and an electrolyte that separates the two poles. When the battery is connected to a device, electrical current flows from the negative terminal to the positive terminal through the device, allowing it to operate. During this process, chemical energy inside the battery is converted into electrical energy.

Hearing aid battery sizes can be identified by the color coding on the packaging. These batteries are usually supplied in blister packs of 6 pieces.
The most common sizes are:
- Size 5 — Red: mini in-ear hearing aids
- Size 10 — Yellow: mini in-ear hearing aids
- Size 13 — Orange: behind-the-ear and in-ear devices
- Size 312 — Brown: in-ear hearing aids
- Size 675 — Blue: behind-the-ear hearing aids**

A LiPo battery should never be deeply discharged. Discharging the battery too far can permanently damage the cells and may cause swelling due to gas formation.
To protect the battery:
- Do not discharge the battery below approximately 3.5 V per cell
- Preferably use only up to around 80% of the battery capacity
- Use a voltmeter or digital battery monitor to check cell voltage
Important: never completely empty a LiPo battery after use, as this can shorten lifespan and create safety risks.

That depends on the battery technology. Older NiCd batteries often needed to be fully discharged before recharging to reduce the memory effect.
For modern battery types such as NiMH, Li-ion, and LiPo, full discharge is no longer necessary and is often discouraged.
Especially with Li-ion and LiPo batteries, deep discharge can damage the battery and may create safety risks. For most modern rechargeable batteries, it is best to recharge them when they are partially discharged rather than fully empty.

Lead-acid batteries should not be deeply discharged or left unused for long periods in a discharged state, as this can permanently damage the battery.
Charging a lead-acid battery typically takes around 14 to 16 hours, which makes this battery technology less suitable for fast charging applications.
For optimal lifespan and performance, regular charging and proper maintenance are important.
Well-known lead-acid battery brands available at Elfa include FIAMM and Yuasa.

The lifespan of a lithium battery depends on several factors, including the battery type, quality, usage conditions, and charging behavior.
Different lithium technologies also have different expected lifespans:
- Lithium-ion (Li-ion) batteries typically last around 2 to 3 years or approximately 300 to 500 charge cycles
- Lithium polymer (LiPo) batteries can often last up to 5 years or exceed 1,000 charge cycles under proper conditions
Over time, all lithium batteries gradually lose capacity, which can result in shorter operating times between charges.
To maximize battery lifespan:
- Follow the manufacturer’s charging recommendations
- Avoid deep discharge
- Avoid excessive heat
- Store batteries under suitable conditions when not in use
Proper charging and maintenance significantly improve battery performance and longevity.

The lifespan of a LiPo battery largely depends on how heavily the battery is used.
When a LiPo battery is frequently operated at maximum load, wear increases significantly and the lifespan may be limited to around 100 charge and discharge cycles.
Under normal or moderate use, a LiPo battery can often achieve up to approximately 400 cycles before performance noticeably declines.
Proper charging, avoiding deep discharge, and preventing overheating can help extend LiPo battery lifespan.

Rechargeable batteries have a limited lifespan and gradually lose capacity over time. As batteries age, they may discharge faster and can also self-discharge more quickly when not in use.
Battery lifespan depends on factors such as:
- Battery chemistry
- Number of charge and discharge cycles
- Depth of discharge
- Charging method
- Operating conditions and temperature
Typical lifespan by battery type
- NiMH batteries: approximately 500 charge cycles under normal use
- Li-ion and LiPo batteries: approximately 400 cycles or around 3 years of average use
- LiFePO4 batteries: typically between 2,000 and 5,000 cycles and often 5 to 10 years of service life
Frequent deep discharge or operating batteries continuously at maximum performance can significantly shorten lifespan.
To maximize battery life:
- Use the correct charger
- Avoid deep discharge where possible
- Store batteries properly
- Avoid excessive heat and overcharging
LiFePO4 batteries are especially known for their exceptionally long lifespan and high cycle stability.

The lifespan of a hearing aid battery depends on both the type of hearing aid and how intensively it is used. In general, larger size 675 batteries can last up to approximately three weeks, while smaller size 5 and 10 batteries often last only a few days. Battery performance is also influenced by factors such as sound settings, the use of wireless features, and the number of hours the hearing aid is used each day.

To charge NiMH batteries safely and correctly, it is important to use a charger specifically designed for NiMH battery technology. This helps prevent overcharging, undercharging, and possible battery damage.
Slow charging is generally recommended, as it reduces heat generation and helps extend battery lifespan.
Well-known brands offering NiMH batteries include GP Batteries, Varta, and Duracell.
For the correct charging of NiMH batteries , it is essential to use a charger specifically designed for NiMH batteries to prevent overcharging or undercharging. Additionally, slow charging is recommended to avoid overheating and damage. Some brands that offer NiMH batteries include GP Batteries, Varta and Duracell.

Battery lifespan depends on several factors, including usage, charging behavior, depth of discharge, and ambient temperature.
To maximize battery life:
- Avoid frequent deep discharging
- Use the correct charger for the battery type
- Charge the battery according to the manufacturer’s recommendations
- Avoid excessive heat
- Store and use the battery at moderate temperatures
In most situations, an ambient temperature of around 20 °C is considered ideal. Higher temperatures can significantly reduce battery lifespan and performance over time. Under normal use and proper charging conditions, a battery will generally last considerably longer.

Proper battery storage helps extend battery lifespan. Storage recommendations differ per battery type:
- Li-ion and LiPo batteries: Store at approximately 50% charge in a cool, dark, and dry place. Never store fully charged or fully discharged batteries.
- Lead-acid batteries: Fully charged AGM and gel batteries can generally be stored unused for up to 6 months at around 20 °C ambient temperature.
- NiCd batteries: Best stored at approximately 40% charge to minimize capacity loss. Under proper conditions, NiCd batteries can often be stored for 3 to 5 years.

NiMH batteries should be charged using a charger specifically designed for NiMH battery technology. These chargers provide a controlled charging current and automatically stop charging once the battery is full, helping to prevent overcharging.
Fast chargers can recharge NiMH batteries within a few hours, but they generate more heat and place additional stress on the battery cells. Standard chargers charge more slowly but are generally better for battery lifespan and long-term performance.
To maximize battery life, it is recommended to fully charge the battery and avoid storing it empty for extended periods. Proper charging helps preserve battery capacity and ensures reliable operation over time.
Elfa supplies both NiMH batteries and reliable chargers for a wide range of applications.
Lighting

ATEX distinguishes different hazardous zones based on the presence of potentially explosive gas or dust atmospheres.
The classification depends on how often and how long an explosive atmosphere may occur in the work environment. These zones determine which equipment and safety systems may be used.
Gas explosion zones
(flammable gas, vapour, or mist mixed with air)
- Zone 0 — Explosive atmosphere is present continuously or for long periods
- Zone 1 — Explosive atmosphere may occur occasionally during normal operation
- Zone 2 — Explosive atmosphere occurs rarely and only for a short time
Dust explosion zones
(combustible dust clouds)
- Zone 20 — Explosive dust atmosphere is continuously present or present for long periods
- Zone 21 — Explosive dust atmosphere may occur occasionally during normal operation
- Zone 22 — Explosive dust atmosphere occurs rarely and only briefly
The lower the zone number, the higher the explosion risk and the stricter the safety requirements for the work environment, equipment, and protection systems.
For more information about ATEX regulations and certified equipment, please visit our ATEX information page.

An ATEX-certified lamp can be identified by the certification markings printed on the lamp housing. These ATEX codes indicate the level of explosion protection and the environments in which the lamp may be safely used.
Common ATEX markings and their meanings
- CE — Confirms that the product complies with European regulations and quality requirements
- Ex — Indicates explosion protection certification
- G / D / GD — Suitable for gas atmospheres (G), dust atmospheres (D), or both (GD)
- EEx / Ex — Approved for use in explosive environments according to European standards
- e / d / i / n — Indicates the protection method used
- IIA / IIB / IIC — Gas group classification, where IIC represents the highest explosion risk
- T1 to T6 — Temperature classification based on maximum surface temperature, where T6 has the lowest surface temperature and highest safety level
ATEX certification must be issued by a notified certification body such as KEMA or DEMKO.
If a lamp is tested by a non-certified inspection body, certification is generally limited to Zone 2 applications, which represents the lowest explosion protection category.
For more information about ATEX regulations and certified lighting solutions, please visit our ATEX information page.

ATEX stands for ATmosphères EXplosibles, referring to potentially explosive atmospheres. If a lamp is ATEX certified, it means the lamp is approved for safe use in hazardous environments where explosive gases or dust may be present.
An ATEX-certified lamp can be recognized by the certification markings printed on the lamp housing.
Elfa offers a range of ATEX-certified lighting solutions from leading brands such as Atexor and Petzl.
Read more about ATEX-certified lighting and explosion-proof equipment here.

Lumens (lm) indicate the total amount of light a lamp produces. It measures the overall light output of the light source, regardless of the environment or distance. A lamp always emits the same number of lumens.
The amount of lumens required depends on factors such as the size and height of the area that needs to be illuminated.
Lux (lx) indicates how much of that light actually reaches a specific surface or area. Lux is measured per square meter and therefore depends on distance, beam angle, and surroundings.
For example: a 10-lumen lamp may illuminate a small cabinet sufficiently, but on a large construction site the lux value becomes too low to provide useful lighting at greater distances.

ATEX is mandatory European legislation for equipment used in explosive atmospheres within the EU, while IECEx is an international voluntary certification system accepted worldwide.
Both systems are based on IEC standards and demonstrate compliance for equipment used in hazardous environments. The main difference is that ATEX applies specifically within the European Union, whereas IECEx is internationally recognized.
Read more about ATEX and explosion-proof equipment here.

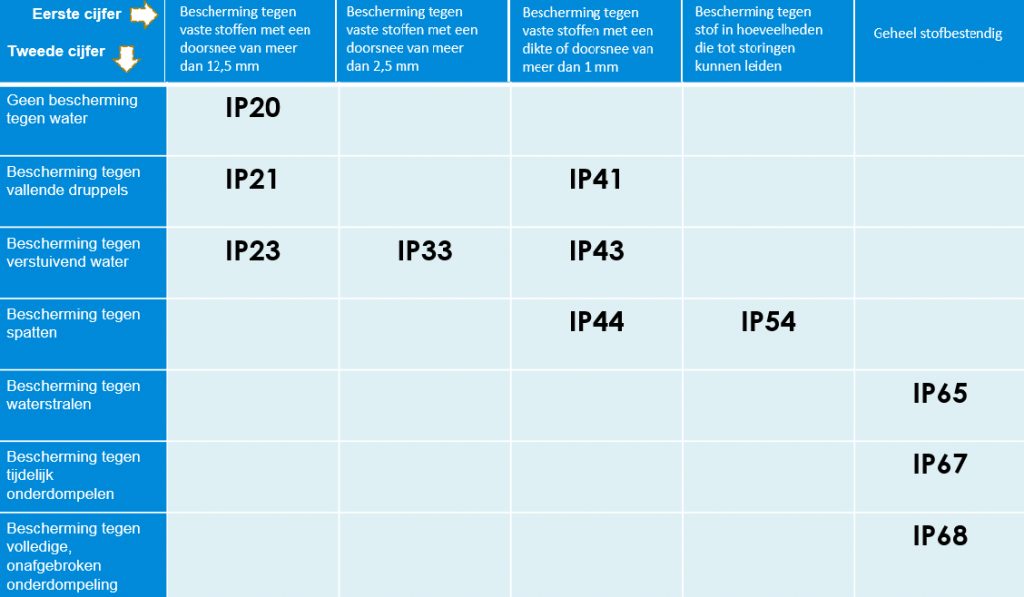
The IP encryption on electricaAn IP rating indicates how well an electrical device is protected against dust, dirt, and moisture in demanding environments.
IP ratings are internationally standardized according to the IEC 60529 standard and consist of two digits:
- The first digit indicates protection against solid objects and dust
- The second digit indicates protection against water and moisture
The higher the number, the greater the level of protection.
Below is an overview of commonly used IP ratings and their meanings.
l devices indicates the degree to which the device is protected against damage caused by use in hostile environments. See below for an overview of commonly used IP designations:

There is a lot of information available. Most of the information is here to find. In addition, below are a number of interesting websites:

There are many different lamp fittings available. These are the most common types:
- Screw fittings — The best-known are E14 (small Edison screw), E27 (standard Edison screw), and E40 (Goliath fitting).
- Push-in fittings — These lamps are inserted directly into the socket using one or more pins.
- TL fittings — Fluorescent tubes use specific fittings depending on the tube type and size.
- Bayonet fittings — These fittings are inserted and twisted so that side pins lock securely into place.

LED stands for Light Emitting Diode and is considered the lighting technology of today and the future.
Unlike traditional incandescent lamps, LEDs do not use a fragile filament. Instead, they convert electrical energy directly into light, making them far more energy-efficient and durable.
Advantages of LED lighting
- Very energy efficient: high light output with low power consumption
- Extremely long lifespan (approximately 20,000 to 100,000 operating hours)
- Resistant to shocks and vibrations because no filament is used
- Suitable for varying temperatures
- Flexible light control using lenses and beam shaping
Possible disadvantages of older LED technology
- Older LED lamps could produce a relatively cool or bluish light color
Modern LED technology now offers a wide range of color temperatures and lighting solutions for both professional and consumer applications.

ATEX stands for ATmosphères EXplosibles, which refers to explosive atmospheres.
An explosive atmosphere is a mixture of flammable gases, vapours, mists, or dust combined with air under atmospheric conditions, where ignition can cause combustion to spread through the entire mixture.
Since July 2003, the European ATEX 95 and ATEX 137 directives have applied. These regulations focus specifically on safety in hazardous environments and require the use of explosion-proof equipment in areas with explosion risks.
ATEX regulations apply to companies and workplaces where flammable substances may create a potentially explosive atmosphere.
Read more about ATEX regulations and explosion-proof equipment here.
Batteries

Emergency lighting must be inspected periodically to ensure it functions properly during a power failure.
According to NEN 1010 and NEN 1838 standards:
- Escape routes must provide at least 1 lux of illumination
- Emergency lighting must operate for at least 1 hour during a power outage
Because batteries gradually lose capacity over time, the maximum recommended battery lifespan is typically 4 years. To remain compliant with regulations and safety standards, replacement is recommended during the 5th year.
Elfa supplies high-temperature NiCd and NiMH emergency lighting batteries that comply with relevant standards and include a 48-month warranty for 1-hour autonomy applications

A parallel connection means connecting the positive terminals together and the negative terminals together. This increases the battery capacity (Ah) while keeping the voltage the same.
A series connection means connecting the positive terminal of one battery to the negative terminal of another battery. This increases the voltage, while the capacity remains the same.
When connecting batteries in parallel or series, always use batteries from the same brand, with the same capacity and production date.
Need both higher voltage and higher capacity? Then batteries can be combined in both series and parallel configurations. In these systems, compensation cables can help balance voltage differences between battery strings for safe and efficient operation.

The lifespan of a lead-acid battery is usually expressed in charge and discharge cycles. In general, the deeper the battery is discharged before recharging, the fewer cycles it can achieve.
On average, lead-acid batteries provide between 300 and 1,000 cycles, depending on the application and usage conditions. In practice, most lead-acid batteries have an expected lifespan of approximately 3 to 5 years.
Proper charging, regular maintenance, and avoiding deep discharge can significantly extend battery life.
Well-known lead-acid battery brands include FIAMM and Yuasa.

The lifespan of a lithium-ion (Li-ion) battery depends on several factors, including the application, usage conditions, and how often the battery is charged and discharged.
Using a suitable charger is essential to maximize battery lifespan and ensure safe charging.
For long-term storage, it is recommended to store Li-ion batteries at approximately 50% charge. Avoid storing batteries fully charged or completely discharged for extended periods.
On average, a lithium-ion battery has a lifespan of around 3 years. Even if the battery is not used intensively, timely replacement is recommended to maintain optimal performance and safety.

Always charge a LiFePO4 battery using a dedicated LiFePO4 battery charger. A fully automatic LiFePO4 charger is easy to use and automatically starts the charging process after connection.
LiFePO4 batteries should preferably not be kept continuously at 100% charge. However, compared to conventional batteries, they can safely handle much deeper discharge levels.
For optimal performance and safety, charge the battery at temperatures between 0 °C and 45 °C. Always follow the maximum charging current, voltage, and temperature limits specified by the charger and battery manufacturer.
Well-known LiFePO4 battery brands include Cellpower and PBQ.

Always use a charger specifically designed for LiFePO4 batteries. These batteries require a dedicated charging profile with the correct voltage and current limits to ensure safe charging and maximize battery lifespan.
Want to know more? Feel free to contact us.

Emergency lighting batteries are often application-specific, which means pricing depends on factors such as battery type, capacity, connector, and autonomy requirements.
In Elfa’s online catalog, you can explore the complete range of emergency lighting batteries and matching connectors, and easily request a quotation for your specific application.

Although LiFePO4 batteries offer many advantages, there are also some disadvantages to consider.
- Higher purchase costs compared to lead-acid batteries and some other lithium-ion technologies
- More limited availability in certain capacities, dimensions, or battery shapes, meaning customization may sometimes be required
- Reduced performance at very low temperatures, which can affect use in cold environments
Despite these limitations, LiFePO4 technology also offers major benefits:
- Maintenance-free operation
- Deep discharge capability up to 100%
- Up to 5 times more charge cycles than lead-acid batteries
- Long lifespan and high safety level
This combination makes LiFePO4 batteries a popular solution for professional and industrial energy storage applications.

LiFePO4 batteries are generally more expensive than conventional batteries. The price depends on factors such as brand, capacity, voltage, and amperage. Entry-level LiFePO4 batteries are available from around 10Ah.
Although the initial investment is higher, LiFePO4 batteries offer a much longer lifespan and retain their capacity better over time compared to traditional battery technologies.
Because of their long cycle life, deep discharge capability, and low maintenance requirements, LiFePO4 batteries are often a cost-effective long-term investment.
Want to know more about LiFePO4 battery prices for your business application? Contact the battery specialists at Elfa.

Lead-acid batteries are among the most widely used rechargeable battery types and offer several important advantages:
- Simple and reliable construction
- Relatively low purchase cost
- Suitable for a wide range of applications
- Proven and widely available technology
Lead-acid batteries are commonly used in:
- Telecommunications
- Data centers
- Railway applications
- Defence systems
- Security and backup power systems
Well-known lead-acid battery brands include FIAMM and Yuasa.

A FIAMM VdS-certified battery is a battery that not only meets standard quality requirements, but also carries an official VdS certification.
This certification is available on selected models within the FIAMM FG series and confirms that the battery complies with strict safety and quality standards.
VdS applies extensive testing and certification procedures to ensure reliable performance and maximum safety, even in demanding conditions and critical applications.
In short, a VdS-certified FIAMM battery offers additional assurance of proven quality, reliability, and safety.

The lead-acid battery is the most commonly used type of rechargeable battery. This is mainly due to its simple construction, reliability, and relatively low cost.
Lead-acid batteries are widely used in applications such as backup power systems, telecommunications, security systems, and industrial equipment.
Visit our lead-acid battery page for more information and an overview of the brands offered by Elfa.

ELP emergency lighting batteries - Elfa Elementenfabriek’s own brand - have a minimum lifespan of 4 years and provide more than 1 hour of autonomy during a power failure.
To ensure maximum reliability and compliance, preventive replacement every 4 years is recommended.

LiFePO4 batteries offer several important advantages compared to conventional battery technologies:
- Lightweight construction
- Fast charging capability
- Low self-discharge
- Long lifespan and high cycle life
- Good resistance to higher temperatures
- More environmentally friendly due to the use of safer materials
- Available in a wide range of capacities and amperages
Well-known LiFePO4 battery brands include Cellpower and PBQ.

A FASTON connection for emergency lighting uses slide-on terminals that are pushed directly onto the battery contacts. This makes the connection robust and quick to replace.
A connector connection uses a dedicated plug that connects directly to the luminaire wiring or circuit board. This helps prevent incorrect installation and connection errors

A lithium iron phosphate (LiFePO4) battery is a lithium-based battery technology that has been used since 1996 and is known for its safety, long lifespan, and stable performance.
Key features of LiFePO4 batteries include:
- Fast charging capability up to 100%
- Higher usable energy storage compared to many lead-acid batteries
- Long cycle life
- High thermal and chemical stability
- Low maintenance requirements
LiFePO4 batteries can typically utilize nearly 100% of their stored energy, while many conventional lead-acid batteries effectively use around 85%.

LiFePO4 (Lithium Iron Phosphate) is a lithium battery technology known for its high safety, long lifespan, and sustainable characteristics.
In a LiFePO4 battery, the cells contain lithium iron phosphate material, which is more environmentally friendly and thermally stable than many other lithium-ion chemistries.
Key advantages of LiFePO4 batteries include:
- Non-flammable chemistry
- High resistance to overheating
- Long cycle life
- High level of operational safety
- Environmentally friendlier materials
- Stable performance over time
Because of these properties, LiFePO4 batteries are widely used in energy storage systems, industrial applications, and electric mobility.

A battery should be replaced when it can no longer hold its charge properly. One common sign is that the battery appears to charge very quickly but also runs empty much faster than before.
This usually indicates that the battery capacity has decreased due to aging and normal use. Over time, all batteries gradually lose capacity and performance.
If you notice reduced runtime, rapid charging, or unstable performance, it is often time to replace the battery.

A LiFePO4 battery looks the same as a conventional battery, but is lightweight, robust, and has high power. The LiFePO4 is a powerful battery that allows you to easily charge all your electronic devices. LiFePO4 batteries do not use toxic materials, thus reducing the risk of fire.
With a LiFePO4 battery, you can use more than 100% of its nominal capacity. In comparison, the usable capacity of a standard battery is between 30% and 50%.



